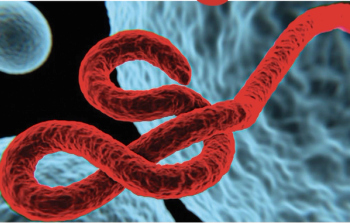
David Wohl, MD, with the Institute for Global Health and Infectious Diseases at the UNC School of Medicine, provides information on this strain, the quarantine protocols, and what the public should know about when the virus sheds and transmissibility of the disease to others.