Gastrointestinal Infections
Latest News

CDC Links Salmonella Outbreak to Metabolic Meals Home Delivery Products

FDA Clears Human Challenge Trial for First Potential Norovirus Antiviral, CDI-988
Video Series

Latest Videos
Shorts

CME Content
More News

Several companies nationwide, with FDA oversight, recalled food products due to potential bacterial contamination, issuing consumer warnings to prevent foodborne illness.
FDA and CDC warn consumers as recalled heat-and-eat meals sold at Kroger and Walmart are tied to multiple hospitalizations, one pregnancy loss, and ongoing risks.

The Centers for Disease Control and Prevention (CDC) provided an update on the outbreak associated with whole fresh cucumbers, which were distributed this spring to stores, restaurants, and other facilities.

Clostridium botulinum and Listeria contamination found in multiple food products, including vegetables, mushrooms, and seafood chowders.

Phase 1b trial data demonstrated immune response, with 85% reduction in viral shedding and robust mucosal immunity in elderly adults.

The company's trivalent vaccine (mRNA-1403) was being studied in a phase 3 trial.
As a result of an independent committee’s determination, the study has been discontinued.

The new version of its molecular test targets the causes of gastroenteritis.

FDA investigates ongoing outbreaks involving contaminated pastries with 18 reported cases of illness across 7 states, and 1 hospitalization.

There are concerns around norovirus risks associated with a certain harvest from Louisiana.

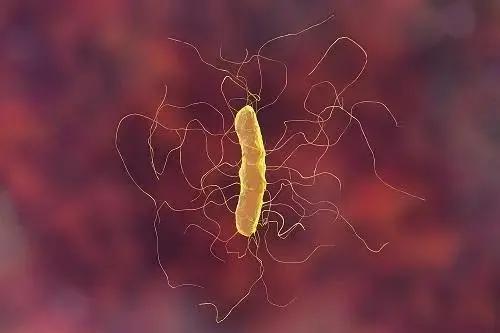
Fecal microbiota transplantation emerges as the top treatment, while probiotics show limited effectiveness in preventing CDI recurrence.

The fully cooked frozen product was not recalled because the product can no longer be purchased. This does apply to consumers who have the product at home.

Sasirekha Ramani, PhD, discusses the potential of 2′FL as a treatment for norovirus and what more research could reveal.
Study shows 2′-fucosyllactose, a sugar in human milk, could reduce norovirus replication, offering hope for new treatments amid rising cases.
Nationwide norovirus cases have recently increased, followed by an FDA warning for consumers to avoid oysters contaminated in a recent outbreak.

From foodborne illness outbreaks to waterborne diseases and beyond, this year saw developments in the gastrointestinal infection space.

A multistate outbreak of E coli O157:H7 infections has been served at catering events, restaurants, and schools.

Most outbreaks were caused by Cryptosporidium, a chlorine-resistant parasite, underscoring the need for stricter safety measures and better facility management.

Sixty-eight people have been infected with this food borne illness across 19 states.

This week, GigaGen has launched a Phase 1 trial for GIGA-2339, a potential functional cure for chronic Hepatitis B, public health concerns like the California raw milk recall due to H5N1 and new findings linking lung dysfunction to cognitive decline in Long COVID patients, and more.

It is not only about cooking the food to the correct temperature, but also how long the food should sit out, and safe storage after the meal is over. Here is more information to have a healthy holiday.

Wolverine Packing Co is recalling ground beef due to potential contamination with E coli O157:H7

Nicole M Iovine discusses the risks associated with this particular E coli strain, challenges posed by organic farming, and offers practical food safety tips to protect vulnerable populations.

Grimmway Farms issued a voluntary recall after 26 out of 27 individuals interviewed as part of the outbreak investigation reported eating their carrots before becoming ill.

Affected products include Taiwan Enoki mushrooms, select soft-ripened cheeses, and Autumn Turkey Sandwiches.