Prevention
Latest News

Video Series

Latest Videos
Podcasts
CME Content
More News

Texas judge Reed O’Connor ruled that requiring employers to cover the medical expense of HIV PrEP is a violation of religious freedom.

The FDA lifted a clinical hold on investigational lenacapavir for HIV treatment and pre-exposure prophylaxis. All clinical studies evaluating injectable lenacapavir can now resume.
Dr. Simon Portsmouth of Shionogi discusses the trial results of their once-daily oral COVID-19 antiviral, S-217622, as well as the significance of the therapy as vaccine efficacy wanes in the age of Omicron.

In Germany, HIV pre-exposure prophylaxis users are recommended to receive regular HIV, STI, and renal function testing. How frequently are PrEP users getting tested?
A large surveillance study of 2 symptom-reporting databases found side effects after Moderna or Pfizer-BioNTech COVID-19 vaccination were frequently nonserious and resolved within 1-2 days.

“People don’t recognize that at least 1 in 5 of the deaths from opioids may result from endocarditis,” said National Institute of Drug Abuse (NIDA) director Dr. Nora Volkow, adding that endocarditis is “100% preventable.”

Convalescent plasma safely raised antibody levels in children at high risk of severe or fatal COVID-19, successfully neutralizing the virus but unlikely to provide lasting protection.

Two different doses of the company's mRNA vaccine were found to increase antibodies by up to 83-fold.
A novel digital pill was 98% effective in tracking daily HIV PrEP adherence.
HPV vaccination decreases cervical cancer incidence and mortality, but it is most effective in young women and girls.

New cohort data from VA medical centers provide more evidence of a correlate between antibody levels and vaccine effectiveness for mRNA vaccines.

The CDC updated its HIV PrEP guidelines, including a recommendation that doctors talk to all sexually active adults about HIV prevention and prescribe PrEP to anyone who asks for it.

Merck announced an enrollment pause in 2 of their stage 3 clinical trials for once-monthly oral islatravir for HIV PrEP. Enrolled participants will continue receiving study medicine.

Only 1 in 3 heterosexually active adults is aware of HIV PrEP, and this is even lower among Hispanic/Latinx men and women.

A new report suggests FDA and CDC deliberations may result in booster doses of the mRNA vaccine becoming available by this weekend.

The Biden administration is also setting mandates for health care workers in facilities receiving federal aid.

The ACIP considered the benefits of the newly emergency-authorized vaccine versus the risks of COVID-19 in children aged 5-11 years old.
29% of cisgender men who have sex with men acquired HIV despite being designated as “low risk.”

Unpublished data from the company's ongoing pediatric trial show children aged 6 to <12 years old have an antibody response greater than that of young adults.
The FDA and CDC have authorized and recommended 3 different booster doses, to be used in "mix-and-match" strategy among eligible adults. What does this mean for immunity?

The decision comes after supporting data from a "mix and match" trial last week, and may align with emergency authorizations for the Moderna and Janssen booster doses.
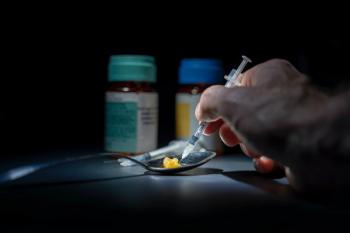
People who inject drugs in rural areas had a less accurate understanding of HIV PrEP, largely due to stigma surrounding sexual orientation and drug use.
The findings have supported the launch of a phase 3 trial called EVERGREEN, which is now underway.
PROVENT Trial: AZD7442 as Long-Acting Pre-Exposure Prophylaxis Protects Against Symptomatic COVID-19
In the primary efficacy analysis, the risk of developing symptomatic COVID 19 was reduced by 77% with AZD7442.

The companies will be seeking emergency authorization for a lighter-dose mRNA vaccine for children aged 5 to <12 years old.