PEN-FAST Forward: Accelerating Penicillin Allergy De-Labeling
PEN-FAST is a validated risk stratification tool that promotes efficient, safe, and effective de-labeling of penicillin allergies.
De-labeling previous penicillin allergies can aid in improving patient outcomes, as well as improve stewardship efforts.
Image credit: SIDP

Why is Penicillin Allergy De-Labeling Important?
Penicillin allergies are the most reported drug reaction in the United States, with up to 10% of people reporting one. However, if these patients undergo allergy testing, up to 95% of them have no reaction.1
We also know that penicillin allergies can cause harm to patients. Alternative treatment options often result in broader spectrum or less effective antibiotic use. In a large cohort study of over 300,000 participants with a mean follow-up time of 6 years, patients with a penicillin allergy had a 14% increase in all-cause mortality compared to patients without a documented allergy.2 Penicillin allergies have been linked to an increase in hospital-acquired infections and resistance, including a 14.1% increase in methicillin-resistant Staphylococcus aureus (MRSA), a 30.1% increase in vancomycin-resistant Enterococcus (VRE), a 23.4% increase in Clostridioides difficile infection, and a 50% increase in surgical site infections.3-4 Suboptimal antibiotic use from documented penicillin allergies has also been shown to increase antibiotic cost, adverse effect rate, length of hospital stay, and readmission rate.1,5
Allergy History
The American Academy of Allergy, Asthma, and Immunology (AAAAI) endorses proactive penicillin allergy de-labeling efforts.6 No matter what de-labeling strategy is employed, it is important to get a thorough and accurate allergy history, including the type of reaction, timing of reaction, any treatment received for the reaction, and antibiotics the patient has tolerated since the reaction.
In patients who have reactions that are more consistent with intolerance than allergy, AAAAI recommends the removal of a penicillin allergy label without any additional testing. In patients whose history is consistent with a true allergy, risk stratification should be done to determine de-labeling strategies. One tool that has been developed and validated to risk stratify patient allergy histories is PEN-FAST.7
PEN-FAST and De-Labeling
Table 1: The PEN-FAST risk stratification tool.
*PEN-FAST is validated only for penicillin allergies. Exclusions for allergy drug challenge apply, including non-IgE mediated reactions and concurrent antihistamines.
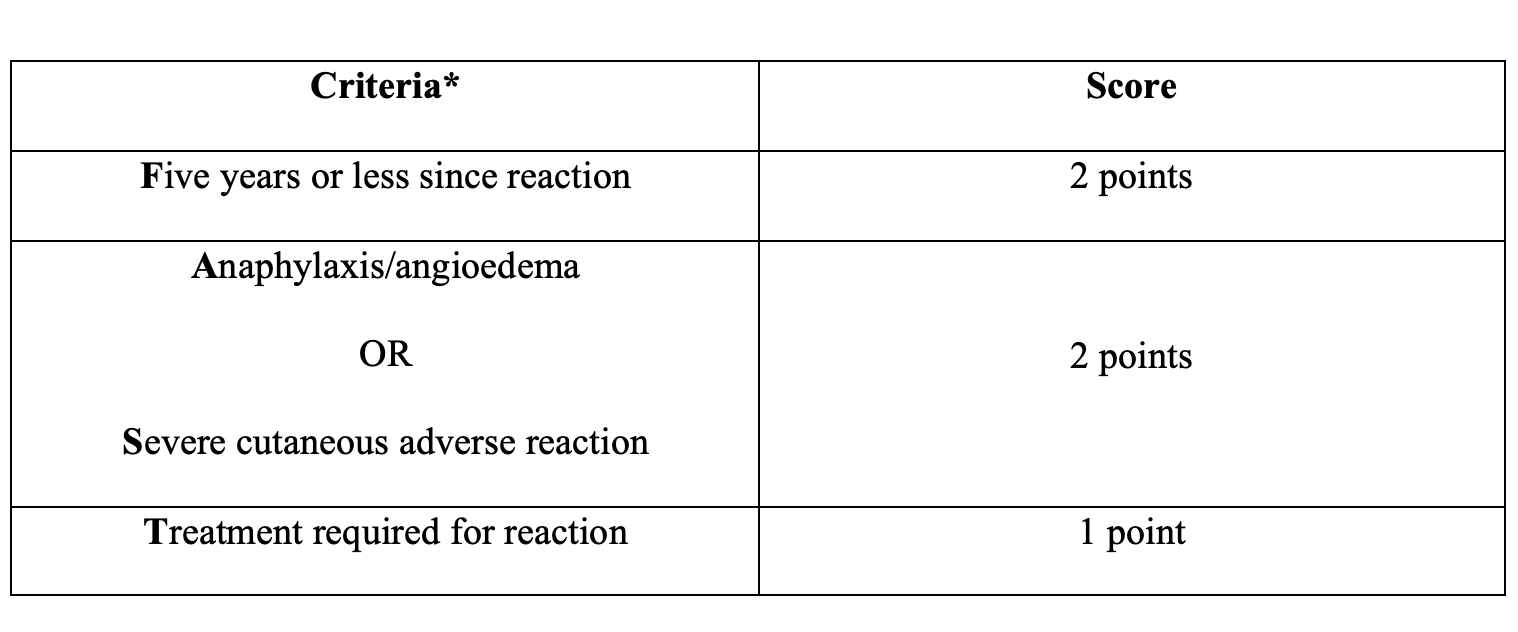
The PEN-FAST score is an incredibly useful tool for determining appropriate de-labeling strategies. It is a relatively short assessment that can be carried out in any health care setting. Among eligible patients that are low-risk (PEN-FAST score of 0-2), an oral amoxicillin challenge may be considered.
Exclusions for an oral amoxicillin challenge in a “low-risk” patient include severe cutaneous adverse reaction (such as Stevens-Johnson syndrome, toxic epidermal necrolysis, or drug reaction with eosinophilia and systemic symptoms), other non-IgE mediated delayed drug reactions such as drug-induced organ damage, or concurrent treatment with antihistamines or high dose steroids.8
During an oral challenge, a single dose of amoxicillin 250 mg is given to the patient, followed by monitoring for 60-120 minutes. If the patient tolerates the dose without a reaction during the monitoring period, the allergy can safely be removed from the patient’s chart and their reaction history should no longer prevent them from receiving penicillins in the future.6
Table 2: PEN-FAST score interpretation
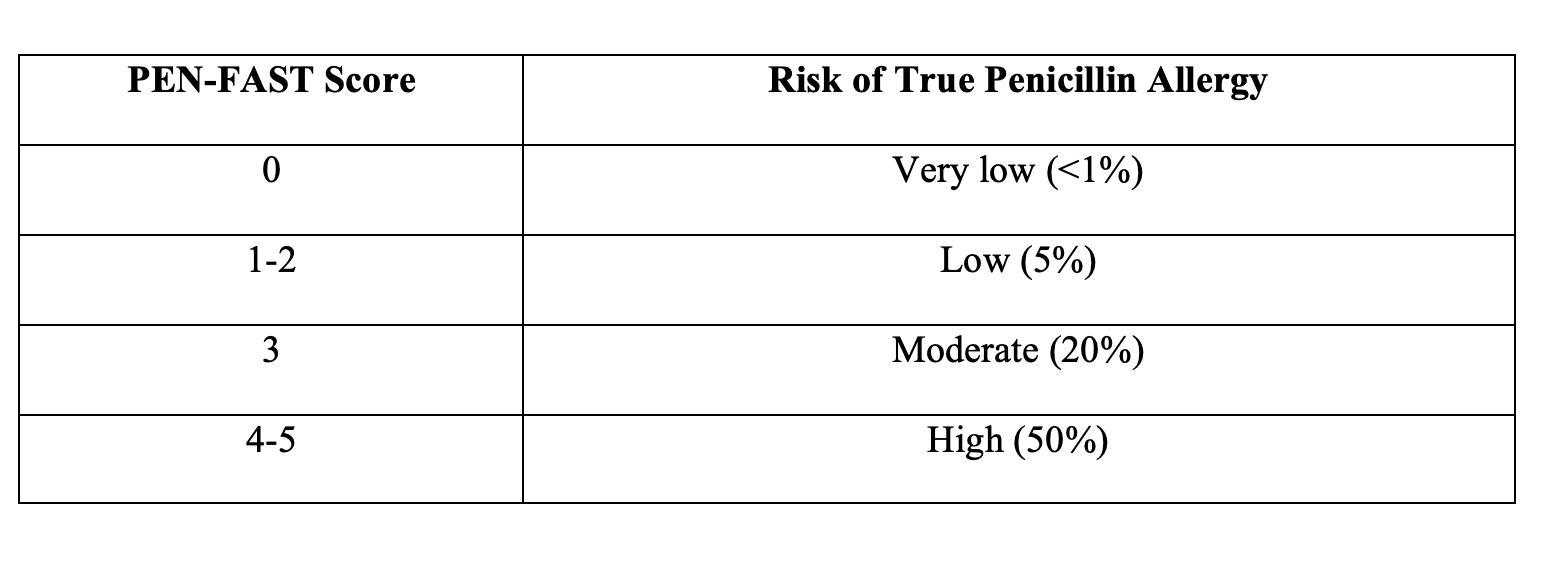
While de-labeling has traditionally been done with a penicillin skin test followed by an oral challenge, the PALACE trial demonstrated that a direct oral challenge in patients with a PEN-FAST score of 0-2 was equally safe to performing skin testing followed by an oral challenge.8 Patients who have moderate or high-risk penicillin allergy history likely require more extensive work-up, including penicillin skin testing, to minimize risk of reaction.
Conclusion
Penicillin allergies are common, and their presence on medical records leads to worse outcomes. Collecting an accurate allergy history and evaluating true allergy risk can help health care professionals identify and remove unnecessary penicillin allergy labels. PEN-FAST is a validated risk stratification tool that can be easily adopted in multiple settings.
References
- Shenoy ES, Macy E, Rowe T, Blumenthal KG. Evaluation and management of penicillin allergy: a review. JAMA. 2019;321(2):188-199. doi:10.1001/jama.2018.19283
- Blumenthal KG, Lu N, Zhang Y, Walensky RP, Choi HK. Recorded penicillin allergy and risk of mortality: a population-based matched cohort study. J Gen Intern Med. 2019;34(9):1685-1687. doi: 10.1007/s11606-019-04991-y
- Macy E, Contreras R. Health care use and serious infection prevalence associated with penicillin “allergy” in hospitalized patients: a cohort study. J Allergy Clin Immunol. 2014;133(3):790-796. doi: 10.1016/j.jaci.2013.09.021
- Blumenthal KG, Ryan EE, Li Y, Lee H, Kuhlen JL, Shenoy ES. The impact of a reported penicillin allergy on surgical site infection risk. Clin Infect Dis. 2018;66(3):329-336. doi: 10.1093/cid/cix794
- Sousa-Pinto B, Blumenthal KG, Macy E, et al. Penicillin allergy testing is cost-saving: an economic evaluation study. Clin Infect Dis. 2021;72(6):924-938. doi: 10.1093/cid/ciaa194.
- Khan DA, Banerji A, Blumenthal KG, et al. Drug allergy: a 2022 practice parameter update. J Allergy Clin Immunol. 2022;150(6):1333-1393. doi: 10.1016/j.jaci.2022.08.028
- Trubiano JA, Vorgin S, Chua KYL. Development and validation of a penicillin allergy clinical decision rule. JAMA Intern Med. 2020;180(5):745-752. doi: 10.1001/jamainternmed.2020.0403
- Copaescu AM, Vogrin S, James F, et al. Efficacy of a clinical decision rule to enable direct oral challenge in patients with low-risk penicillin allergy: the PALACE randomized clinical trial. JAMA Intern Med. 2023;183(9):944-952. doi: 10.1001/jamainternmed.2023.2986
Newsletter
Stay ahead of emerging infectious disease threats with expert insights and breaking research. Subscribe now to get updates delivered straight to your inbox.
