In the final episode, the panelists offer some insights around following the federal agencies' ever-changing vaccine policies and the value of following professional medical societies' guidance during this period of uncertainty.

In the final episode, the panelists offer some insights around following the federal agencies' ever-changing vaccine policies and the value of following professional medical societies' guidance during this period of uncertainty.
The DOTS trial results found that although dalbavancin did not demonstrate superiority over standard intravenous therapy for complicated Staphylococcus aureus bacteremia, it was noninferior in efficacy, with similar safety, and offers a convenient 2-dose alternative that may reduce OPAT-related complications and health care utilization in select patients.

In this episode, the panelists discuss the potential concerns around changing vaccine guidelines and the importance of the American Academy of Pediatrics (AAP) stepping in to offer their recommendations.

The Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices (ACIP) was supposed to convene this week to discuss COVID-19 vaccines. However, a district court’s decision to block recent vaccine policy changes has delayed the committee's meeting amid ongoing controversy over immunization guidance and rising public health concerns such as measles outbreaks. Here is some commentary on the inner workings of the current ACIP.

A district court judge has ruled against the current administration's vaccine policies and throws into jeopardy this week’s Advisory Committee on Immunization Practices meetings that were scheduled to discuss COVID-19 vaccines.

In this episode, the panelists discuss the probability that with vaccine recommendations changing across states and federally, costs could shift to families and increase health disparities.

The World Health Organization has released 3 new target product profiles aimed at guiding the development of urgently needed antibiotics for severe drug-resistant infections, including multidrug-resistant gram-negative infections, resistant gram-positive infections in vulnerable patients, and bacterial meningitis.

A phase 3 clinical trial found the Butantan-DV tetravalent dengue vaccine provided 80.5% protection against severe dengue with warning signs over 5 years and prevented hospitalizations among vaccinated participants.

This week, read about tapering vancomycin for 2 weeks after a 2-week standard dose treatment of Clostridioides difficile, phage-antibiotic combinations, another episode from our vaccine roundtable, and more.

For those who achieve the DCLS degree, they can expand their career beyond the laboratory, bringing their expertise to the table to aid clinicians with consultation, improve patient outcomes, and increase health system efficiency. Brandy Gunsolus, DCLS, discusses the motivation that led her to become the first person in the country to achieve this newer accreditation.

The latest CDC numbers show a 6.3% increase of measles cases from week-to-week. This is a decrease in the number of cases from last week where there was a 12.76% increase.

In this episode, the panelists talk about how they broach conversations with expectant mothers around counseling on vaccinations for their babies.

The FDA’s Vaccines and Related Biological Products Advisory Committee (VRBPAC) unanimously voted for a trivalent vaccine composition for use in the US, protecting against influenza A (H1N1 and H3N2) and influenza B (Victoria lineage).
Tapering vancomycin over 2 weeks after a 2-week full dose regimen for Clostridioides difficile reduced the rate of infection recurrence.

An increase in cases of blastomycosis in upstate New York over the past decade suggests it is becoming endemic, although it is not yet a reportable infection.
Peptilogics has enrolled the first patient in its RETAIN trial evaluating PLG0206, a first-in-class anti-biofilm therapy designed to treat prosthetic joint infections.

In this episode, the panelists discuss how the pediatric immunization schedule changes could potentially influence vaccines' inequities, and how vaccine education continues to play a significant role in immunization uptake and improving health equity.

A phase 2 trial published in The Lancet Infectious Diseases found that the investigational antibiotic CRS3123 achieved high clinical cure rates comparable to vancomycin while significantly reducing recurrence and preserving the gut microbiome in patients with Clostridioides difficile infection.

In this episode, the panel discusses the continued importance of getting all children vaccinated and the potential unintended consequences of fewer individuals being immunized if the current trends continue.

Emerging preclinical, compassionate-use, and randomized trial data suggest phage-antibiotic combinations may benefit select patients with multidrug-resistant infections, yet critical gaps in standardization and evidence remain.

The rate of HBV vaccination of newborns in the US has declined by over 10% in the past 2 years, reversing decades of increasing coverage.

This week, read about the latest measles numbers, a risk assessment and antibiotic treatment of GAS pharyngitis, an analysis of ceftolozane-tazobactam over aminoglycosides/polymyxins for resistant P aeruginosa, and more.

The FDA’s biologics center faces another leadership shake-up as Vinay Prasad, MD, MPH, plans an exit in April, leaving questions over vaccine rules and gene-therapy approvals.

The latest CDC numbers show a 12.76% increase of measles cases from week-to-week. This is a slight decrease in the number of cases from last week where there was a 15.7% increase. The data also shows outbreaks in 3 new states.

Despite increased risk for severe outcomes, most older adults and those with comorbidities do not receive antiviral treatment for COVID-19.

Despite the absence of vaccine data to support making changes, the ACIP continues to alter the pediatric schedule that will see less children getting immunized against vaccine-preventable diseases.
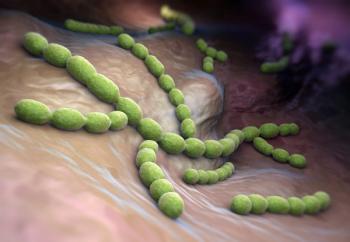
In 2025, the Infectious Diseases Society of America published the first part of an update to the clinical practice guideline on the diagnosis and management of GAS pharyngitis. Here is an overview of the risk assessment and antibiotic treatment of GAS pharyngitis.

In this episode of our roundtable discussion, the panelists talk about the changes to the CDC’s ACIP meetings.

By launching during the Super Bowl pregame and the Grammy Awards, Gilead’s One2PrEP campaign used music, mainstream media, and bold visibility to normalize conversations about HIV prevention and expand awareness of evolving PrEP options.

As the US continues to undergo massive policy changes to the childhood vaccine schedule, clinicians and families are left uncertain on recommendations and guidance. Our roundtable looks to provide some insights and answer questions to some of the more pressing topics about this evolving issue.