
This is the first in a series of discussions between medical peers around RSV vaccines and specifically the newly approved Moderna RSV vaccine for seniors.

This is the first in a series of discussions between medical peers around RSV vaccines and specifically the newly approved Moderna RSV vaccine for seniors.

The federal agency’s decision expands the vaccine’s eligibility to people 50 years of age and older, making it the first one for this age group.

Ongoing surveillance from the CDC and FDA will continue to evaluate the risk of Guillan-Barré syndrome to guide further ACIP recommendations.

The company’s mRNA 1345 vaccine was given the nod by the federal agency and is indicated for seniors for the prevention of RSV-associated lower respiratory tract disease (RSV-LRTD).

The program allows participants to reserve doses and be eligible for priority shipping, among other benefits.

The federal agency said that due to administrative constraints, they will not complete the review of mRNA-1345 by the PDUFA date of May 12.
The investigational vaccine, mRNA-1345, developed for seniors has its FDA PDUFA date in a few days.

In a new real-world study, Sanofi and AstraZeneca’s nirsevimab-alip (Beyfortus) demonstrated high efficacy against respiratory syncytial virus (RSV) lower respiratory tract disease (LRTD).

FDA-approves mpox vaccine, FDA grants EUA for at-home multiplex papid test, FDA grants PDUFA for GSK 5-in-1 meningococcal vaccine, and more this month from the FDA.
The study findings imply newly available prevention strategies for which older children—ages 2 to 5 years—are not currently eligible should be prioritized.

The company is going to submit their data to the FDA to seek approval for people within this age group.
Evaluating the importance of RSV vaccination implications for adults.

An FDA decision on the company’s mRNA-1345 is slated for May 12.

The RSV prefusion F protein–based maternal vaccine phase 3 trial was halted due to the risk of this adverse event.

Impact of the RSVPreF3 vaccine on respiratory illness in older adults in the US.
A new strategy for universal infant immunization in the 2023-2024 RSV season faces implementation and medication access barriers.
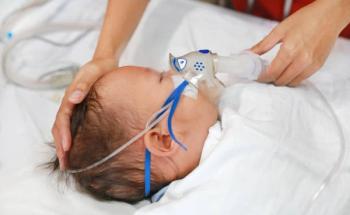
High efficacy of Nirsevimab in preventing RSV hospitalizations was revealed.
Though a small number of infants received nirsevimab in the analysis, results support existing nirsevimab recommendations to prevent serious RSV disease in infants.

A new study shows that, among the tridemic viruses, RSV was the leading cause of pediatric hospitalizations.
Changes encompass clarification of recommendations for scheduled vaccinations and the addition of the RSV vaccines for the first time.
The panel discusses the importance of discussing the immunizations with patients now, as well as the value of them in future seasons.

In the fourth installment of RSV Roundtable, our panel explains the challenges in protecting the senior population against RSV.

In the third installment of RSV Roundtable, our panel discusses the impact nirsevimab has had this RSV season.

In the second installment of the RSV Roundtable, our panel explains how they are educating patients and parents when it comes to RSV, vaccines and preventive measures, and limited treatment availability.

Last year’s historic RSV season left many wondering if this was going to become a recurring trend. Physicians weigh in on what they are seeing at their institutions in terms of infection rates.

A clinician discusses the vaccines' efficacy, which risk factors may rank higher for disease severity, and offers insights on counseling patients.
The FDA and CDC announced the availability of additional doses of nirsevimab-alip for infants at high-risk of infection.

Two RSV vaccines for seniors were FDA approved this year and here is an overview of the vaccines and the study data.

Tina Tan, MD, FAAP, FIDSA, FPIDS, discusses the benefits of having new tools to fight RSV in infants, potential vaccine hesitancy, and RSV trends currently being observed.

The federal agency says the data shows immunization reduces hospitalizations by more than 57 percent in first 6 months after birth.