
TAXIS Pharmaceuticals Chief Scientific Officer Ajit Parhi, PhD, offers some insights on the disease burden, and an overview of his company’s pipeline around developing a therapy for this STI.

TAXIS Pharmaceuticals Chief Scientific Officer Ajit Parhi, PhD, offers some insights on the disease burden, and an overview of his company’s pipeline around developing a therapy for this STI.

In the final installment of the short series around fungal infections, Jatin Vyas, PhD, MD, discusses the challenges of developing antifungals.

The organization announced this year's event, which aims to bring together stakeholders in Washington, DC, to advance awareness, policy, and patient-centered solutions for Clostridioides difficile infection.

In the second installment of the short series around fungal infections, Jatin Vyas, PhD, MD, discusses the increase in Candida auris including its unique clades and past challenges in diagnosing this difficult-to-treat infection.
In 2018, the FDA approved omadacycline as a new oral/intravenous third generation tetracycline to treat Community Acquired Bacterial Pneumonia (CABP) and Acute Bacterial Skin and Skin Structure Infections (ABSSSI). Here is a clinical overview of omadacycline as well as recent phase 2 clinical trial results favoring oral omadacycline as an oral treatment option for Mycobacterium abscessus pulmonary disease.

Jatin Vyas, PhD, MD, discusses where the research is headed as it takes a page from the oncology world in trying to harness the immune system and combining with fungal therapeutics to treat these difficult infections.

Martin Gartland, PhD, discusses early data on the investigational therapy, VH184, including the potential for long-acting treatment and looking to address emerging drug resistance challenges.

This week, read about a new COVID variant, the test and treat paradigm model for hepatitis C, and a mission around bringing Americans with Ebola back to the US.

The latest CDC numbers show a 5.92% increase of measles cases from week-to-week. This is a decrease in the percentage of cases from last week where there was a 9.18% increase.

Despite the federal government’s recent stance on pandemic planning, author Kevin Hazzard reminds us of the importance of public health work in this area.

In this episode, author Kevin Hazzard talks about the preflight logistics as well as other issues that needed to be corrected for successful transatlantic flights.

Scynexis has secured FDA Qualified Infectious Disease Product (QIDP) and Fast Track designations for its next-generation antifungal SCY-247, underscoring the drug’s potential to address the escalating global threat of multidrug-resistant fungal infections such as Candida auris.

In our second episode of our series, author Kevin Hazzard discusses the measures needed to protect the pilots and medical staff on board the plane from contracting Ebola.

A small airline company with a cowboy mentality stepped up to become the first aviation company to fly American patients back to the US to receive treatment.

A newly identified SARS-CoV-2 variant, BA32, is spreading globally with mutations that may reduce vaccine and prior infection protection, underscoring the continued importance of genomic surveillance.
New research from UC San Francisco (UCSF) suggests hepatitis C cure may be reliably confirmed as early as four weeks after treatment, potentially improving care access for underserved populations.

A phase 3 trial of an investigational Lyme disease vaccine from Pfizer Inc. and Valneva SE demonstrated more than 70% efficacy, signaling potential progress toward the first approved human vaccine for the disease. Pfizer is planning to move forward with regulatory submissions for potential approval.
A new meta-analysis finds alarming increases in colistin resistance among Acinetobacter baumannii and Pseudomonas aeruginosa across Africa, threatening the effectiveness of one of the last-resort antibiotics.

This week, read about the DOTs trial, the latest measles numbers, the World Health Organization's new targets for antibiotic development, and more.

The latest CDC numbers show a 9.18% increase of measles cases from week-to-week. This is an increase in the number of cases from last week where there was a 6.3% increase.

Advocates are pushing for federal action to ensure copay assistance counts toward patient deductibles, as insurers’ use of accumulator programs continues to drive up out-of-pocket costs. Carl Schmid, executive director of the HIV+Hepatitis Policy Institute, offers commentary to this unresolved issue.

In this month's column, we look at the leadership change at CDC, an amicus brief supporting the AAP's lawsuit against HHS changes to the pediatric vaccine schedule, and potential clinical repercussions around the reports the federal government is considering removing the COVID-19 vaccine from the market.

A new bipartisan bill aims to eliminate cost barriers and expand access to PrEP by mandating comprehensive coverage and launching a national program for uninsured populations.
In the final episode, the panelists offer some insights around following the federal agencies' ever-changing vaccine policies and the value of following professional medical societies' guidance during this period of uncertainty.
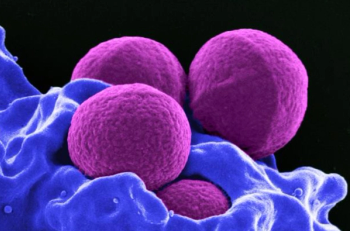
The DOTS trial results found that although dalbavancin did not demonstrate superiority over standard intravenous therapy for complicated Staphylococcus aureus bacteremia, it was noninferior in efficacy, with similar safety, and offers a convenient 2-dose alternative that may reduce OPAT-related complications and health care utilization in select patients.

In this episode, the panelists discuss the potential concerns around changing vaccine guidelines and the importance of the American Academy of Pediatrics (AAP) stepping in to offer their recommendations.

The Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices (ACIP) was supposed to convene this week to discuss COVID-19 vaccines. However, a district court’s decision to block recent vaccine policy changes has delayed the committee's meeting amid ongoing controversy over immunization guidance and rising public health concerns such as measles outbreaks. Here is some commentary on the inner workings of the current ACIP.

A district court judge has ruled against the current administration's vaccine policies and throws into jeopardy this week’s Advisory Committee on Immunization Practices meetings that were scheduled to discuss COVID-19 vaccines.

In this episode, the panelists discuss the probability that with vaccine recommendations changing across states and federally, costs could shift to families and increase health disparities.

The World Health Organization has released 3 new target product profiles aimed at guiding the development of urgently needed antibiotics for severe drug-resistant infections, including multidrug-resistant gram-negative infections, resistant gram-positive infections in vulnerable patients, and bacterial meningitis.