Respiratory Infections
Latest News

Latest Videos

More News
The intervention included meeting with experts, texting services to improve and facilitate communication, and data collection and dissemination.

The CDC is urging all eligible persons to receive a flu shot, but a new survey shows fewer adults are planning to get the vaccine this season.

Catch up on Contagion's top stories from the month of September.

The federal agency recommends using either PCV13 or PCV15 as part of a 4-dose series for pediatric patients between the ages of 2-59 months.
Eravacycline is an antibiotic that has been touted for possible treatment of difficult-to-treat resistant (DTR) Gram-negative infections. But can it tackle carbapenem-resistant Acinetobacter baumannii (CRAB) infections?
Why do some children develop polio-like paralysis after contracting enterovirus? Here’s everything we know about the rare but increasing reports of acute flaccid myelitis.
COVID-19 viral load rebound occurred in 2.3% of nirmatrelvir–ritonavir (Paxlovid) recipients and in 1.7% of placebo recipients.
A new study found that patients hospitalized with COVID-19 are at significantly higher risk for venous thromboembolism in the ensuing 3 months than are patients hospitalized with influenza.

Pfizer-BioNTech and Moderna have developed new booster shots optimized for Omicron. They could be approved as soon as Labor Day.

From its results, the company plans to file a Biologics License Application (BLA) with the FDA.

The incubation period of the original, wild-type COVID-19 strain was 6.65 days, while the Omicron variant's incubation period has shortened to 3.42 days.
56% of adults with evidence of Omicron seropositivity did not know they were infected.

CDC links 2021 outbreak of tuberculosis in US affecting 113 persons and causing at least 3 deaths to bone grafts from one infected, deceased donor.

The CDC is advising clinicians to test for parechovirus in infants exhibiting fever, sepsis-like syndrome, or signs of neurological impairment.

Children and adolescents previously infected with COVID-19 or MIS-C demonstrated insufficient antibody titers and neutralization capability against the Omicron variant.

At 12 months after COVID-19 infection, anti-SARS-CoV-2 spike receptor-binding domain IgG persisted in all age groups.
Investigational vaccine demonstrates efficacy against respiratory syncytial virus in trial with adults inoculated with active RSV.
Antibody levels decline in individuals with hybrid immunity similarly to vaccinated individuals, meaning all people need a booster vaccine to protect against future, potentially severe, infection.

Maternal mortalities increased 33.3% during the COVID-19 pandemic, while overall deaths increased 22%. Excess deaths were most prevalent in Hispanic and Black mothers.

Many people who self-report as symptomatic for COVID-19 neglect to get tested, largely citing not knowing where to go for a test as the reason.
With the impending start to the 2022-2023 influenza season just a few months away, here is some information on what is being studied for vaccines as well as recommendations and new testing.
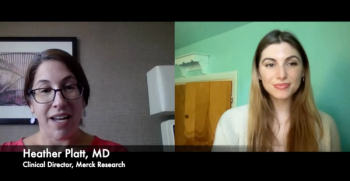
Dr. Heather Platt discusses the process of developing V116, a pneumococcal vaccine that targets the serotypes most prevalent in those 65 years and older.

Women who were taking hormone replacement therapy (HRT) for at least 6 months before COVID-19 infection had a 22% reduced risk of mortality.
Studies, advances, and authorizations continue in this paramount area within infectious disease.

Out of 102 monoclonal antibodies tested, only Cv2.1169 and Cv2.3194 cross-neutralized all variants of concern, including Omicron BA.1 and BA.2 subvariants.